Detection of Catalyst Poisons
Organic molecules, sulphur species and other contaminants in the feed, reactants or intermediate products within chemical processes can affect the activity/efficiency of the costly catalysts even at trace level. Thus there is a strong need to monitor and remove smallest concentrations of them.
A customized version of the GC-IMS made by G.A.S. has proven to be a very useful online monitoring tool to test for a variety of above mentioned species 24/7 on-site at low ppb-level.
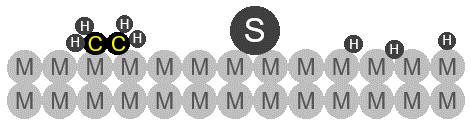
Schematic drawing of sulfur poisoning in ethene (CH4) hydrogenation process. A sulfur atom (S) strongly binds to the catalyst (M) metal surface, reducing the catalytic activity by the following exemplary effects:
-
Sterically blocking adsorption/reaction sites of the catalyst (also inhibiting lateral diffusion)
-
Electronically modification of catalyst surface
Taken from [4]. Copyright 2006, Wiley-Interscience.
[4] Nelson, A.E. (2007), Fundamentals of Industrial Catalytic Processes, 2nd Edition. C. H. Bartholomew and Robert J. Farrauto John Wiley and Sons, Hoboken, NJ, 966 pp., 2006. Can. J. Chem. Eng., 85: 127-128.

 Deutsch
Deutsch English
English Chinese
Chinese